
Browsing Toxins
Displaying toxin 826 - 850 of 3678 in total
| T3DB ID | Name CAS Number | Formula Weight | Structure | Type | Mechanism of Toxicity |
|---|---|---|---|---|---|
| T3D1001 | Propamocarb 24579-73-5 | C9H20N2O2 188.267 g/mol |  |
| Propamocarb is a cholinesterase or acetylcholinesterase (AChE) inhibitor. Carbamates form unstable complexes with chlolinesterases by carbamoylation of the active site...more Number of Targets: 2 |
| T3D1002 | Propham 122-42-9 | C10H13NO2 179.216 g/mol |  |
| Propham is a cholinesterase or acetylcholinesterase (AChE) inhibitor. Carbamates form unstable complexes with chlolinesterases by carbamoylation of the active sites of...more Number of Targets: 2 |
| T3D1003 | Propoxur 114-26-1 | C11H15NO3 209.242 g/mol | 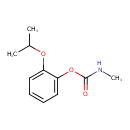 |
| Propoxur is a cholinesterase or acetylcholinesterase (AChE) inhibitor. Carbamates form unstable complexes with chlolinesterases by carbamoylation of the active sites o...more Number of Targets: 13 |
| T3D1004 | Proximphan 2828-42-4 | C10H12N2O2 192.215 g/mol |  |
| Proximphan is a cholinesterase or acetylcholinesterase (AChE) inhibitor. Carbamates form unstable complexes with chlolinesterases by carbamoylation of the active sites...more Number of Targets: 2 |
| T3D1005 | Pyrolan 87-47-8 | C13H15N3O2 245.277 g/mol |  |
| Pyrolan is a cholinesterase or acetylcholinesterase (AChE) inhibitor. Carbamates form unstable complexes with chlolinesterases by carbamoylation of the active sites of...more Number of Targets: 2 |
| T3D1006 | Sulfallate 95-06-7 | C8H14ClNS2 223.786 g/mol |  |
| The metabolic products of triallate, diallate and sulfallate appear to be mutagenic or carcinogenic. In particular, the 2-chloro-allyl group is responsible for the mu...more Number of Targets: 3 |
| T3D1007 | Sulfocarbathione 114654-31-8 | C5H9NO2S3 211.325 g/mol |  |
| Sulfocarbathione is a cholinesterase or acetylcholinesterase (AChE) inhibitor. Carbamates form unstable complexes with chlolinesterases by carbamoylation of the active...more Number of Targets: 2 |
| T3D1008 | Swep 1918-18-9 | C8H7Cl2NO2 220.053 g/mol |  |
| Swep is a cholinesterase or acetylcholinesterase (AChE) inhibitor. Carbamates form unstable complexes with chlolinesterases by carbamoylation of the active sites of th...more Number of Targets: 2 |
| T3D1009 | Tazimcarb 40085-57-2 | C8H13N3O3S 231.272 g/mol |  |
| Tazimcarb is a cholinesterase or acetylcholinesterase (AChE) inhibitor. Carbamates form unstable complexes with chlolinesterases by carbamoylation of the active sites ...more Number of Targets: 2 |
| T3D1010 | Terbucarb 1918-11-2 | C17H27NO2 277.402 g/mol |  |
| Terbucarb is a cholinesterase or acetylcholinesterase (AChE) inhibitor. Carbamates form unstable complexes with chlolinesterases by carbamoylation of the active sites ...more Number of Targets: 2 |
| T3D1011 | Thiobencarb 28249-77-6 | C12H16ClNOS 257.780 g/mol |  |
| Thiobencarb is a cholinesterase or acetylcholinesterase (AChE) inhibitor. Carbamates form unstable complexes with chlolinesterases by carbamoylation of the active site...more Number of Targets: 16 |
| T3D1012 | Thiocarboxime 25171-63-5 | C7H11N3O2S 201.246 g/mol |  |
| Thiocarboxime is a cholinesterase or acetylcholinesterase (AChE) inhibitor. Carbamates form unstable complexes with chlolinesterases by carbamoylation of the active si...more Number of Targets: 2 |
| T3D1013 | Thiodicarb 59669-26-0 | C10H18N4O4S3 354.469 g/mol |  |
| Thiodicarb is a cholinesterase or acetylcholinesterase (AChE) inhibitor. Carbamates form unstable complexes with chlolinesterases by carbamoylation of the active sites...more Number of Targets: 17 |
| T3D1014 | Thiofanox 39196-18-4 | C9H18N2O2S 218.316 g/mol |  |
| Thiofanox is a cholinesterase or acetylcholinesterase (AChE) inhibitor. Carbamates form unstable complexes with chlolinesterases by carbamoylation of the active sites ...more Number of Targets: 2 |
| T3D1015 | Tillam sulfoxide 51892-60-5 | C10H21NO2S 219.344 g/mol |  |
| Some thiocarbamates (EPTC, Molinate, Pebulate, and Cycloate) share a common mechanism of toxicity, i.e. the inhibition of acetylcholinesterase. An acetylcholinesterase...more Number of Targets: 2 |
| T3D1016 | Tiocarbazil 36756-79-3 | C16H25NOS 279.441 g/mol |  |
| Some thiocarbamates (EPTC, Molinate, Pebulate, and Cycloate) share a common mechanism of toxicity, i.e. the inhibition of acetylcholinesterase. An acetylcholinesterase...more Number of Targets: 2 |
| T3D1017 | Triallate 2303-17-5 | C10H16Cl3NOS 304.664 g/mol |  |
| The metabolic products of triallate, diallate and sulfallate appear to be mutagenic or carcinogenic. In particular, the 2-chloro-allyl group is responsible for the mu...more Number of Targets: 9 |
| T3D1018 | Trimethacarb 2686-99-9 | C11H15NO2 193.242 g/mol |  |
| Trimethacarb is a cholinesterase or acetylcholinesterase (AChE) inhibitor. Carbamates form unstable complexes with chlolinesterases by carbamoylation of the active sit...more Number of Targets: 2 |
| T3D1020 | Vernolate 1929-77-7 | C10H21NOS 203.345 g/mol |  |
| Some thiocarbamates (EPTC, Molinate, Pebulate, and Cycloate) share a common mechanism of toxicity, i.e. the inhibition of acetylcholinesterase. An acetylcholinesterase...more Number of Targets: 4 |
| T3D1021 | Xylylcarb 2425-10-7 | C10H13NO2 179.216 g/mol |  |
| Xylylcarb is a cholinesterase or acetylcholinesterase (AChE) inhibitor. Carbamates form unstable complexes with chlolinesterases by carbamoylation of the active sites ...more Number of Targets: 2 |
| T3D1022 | Telodrin 297-78-9 | C9H4Cl8O 411.751 g/mol |  |
| It antagonizes the action of the neurotransmitter gamma-aminobutyric acid (GABA) acting at the GABA-A receptors, effectively blocking the GABA-induced uptake of chlori...more Number of Targets: 38 |
| T3D1023 | Isodrin 465-73-6 | C12H8Cl6 364.910 g/mol |  |
| It antagonizes the action of the neurotransmitter gamma-aminobutyric acid (GABA) acting at the GABA-A receptors, effectively blocking the GABA-induced uptake of chlori...more Number of Targets: 38 |
| T3D1024 | Allethrin 584-79-2 | C19H26O3 302.408 g/mol |  |
| Both type I and type II pyrethroids exert their effect by prolonging the open phase of the sodium channel gates when a nerve cell is excited. They appear to bind to th...more Number of Targets: 36 |
| T3D1025 | Cismethrin 35764-59-1 | C22H26O3 338.440 g/mol | 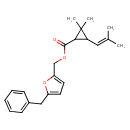 |
| Both type I and type II pyrethroids exert their effect by prolonging the open phase of the sodium channel gates when a nerve cell is excited. They appear to bind to th...more Number of Targets: 19 |
| T3D1026 | Phenothrin 26002-80-2 | C23H26O3 350.451 g/mol |  |
| Both type I and type II pyrethroids exert their effect by prolonging the open phase of the sodium channel gates when a nerve cell is excited. They appear to bind to th...more Number of Targets: 21 |