| Record Information |
|---|
| Version | 2.0 |
|---|
| Creation Date | 2009-07-21 20:28:30 UTC |
|---|
| Update Date | 2014-12-24 20:25:55 UTC |
|---|
| Accession Number | T3D3003 |
|---|
| Identification |
|---|
| Common Name | Chloroprocaine |
|---|
| Class | Small Molecule |
|---|
| Description | Chloroprocaine hydrochloride is a local anesthetic given by injection during surgical procedures and labor and delivery. Chloroprocaine, like other local anesthetics, blocks the generation and the conduction of nerve impulses, presumably by increasing the threshold for electrical excitation in the nerve, by slowing the propagation of the nerve impulse and by reducing the rate of rise of the action potential. |
|---|
| Compound Type | - Amine
- Anesthetic, Local
- Drug
- Ester
- Ether
- Metabolite
- Organic Compound
- Organochloride
- Synthetic Compound
|
|---|
| Chemical Structure | |
|---|
| Synonyms | | Synonym | | 2-Chloroprocaine | | 4-amino-2-Chlorobenzoic acid 2-(diethylamino)ethyl ester | | Chloroprocain | | Chloroprocainum | | Chlorprocaine | | Cloroprocaina | | Nesacaine | | Nesacaine-CE | | Piocaine |
|
|---|
| Chemical Formula | C13H19ClN2O2 |
|---|
| Average Molecular Mass | 270.755 g/mol |
|---|
| Monoisotopic Mass | 270.114 g/mol |
|---|
| CAS Registry Number | 133-16-4 |
|---|
| IUPAC Name | 2-(diethylamino)ethyl 4-amino-2-chlorobenzoate |
|---|
| Traditional Name | chloroprocaine |
|---|
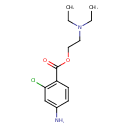
| SMILES | CCN(CC)CCOC(=O)C1=C(Cl)C=C(N)C=C1 |
|---|
| InChI Identifier | InChI=1S/C13H19ClN2O2/c1-3-16(4-2)7-8-18-13(17)11-6-5-10(15)9-12(11)14/h5-6,9H,3-4,7-8,15H2,1-2H3 |
|---|
| InChI Key | InChIKey=VDANGULDQQJODZ-UHFFFAOYSA-N |
|---|
| Chemical Taxonomy |
|---|
| Description | belongs to the class of organic compounds known as benzoic acid esters. These are ester derivatives of benzoic acid. |
|---|
| Kingdom | Organic compounds |
|---|
| Super Class | Benzenoids |
|---|
| Class | Benzene and substituted derivatives |
|---|
| Sub Class | Benzoic acids and derivatives |
|---|
| Direct Parent | Benzoic acid esters |
|---|
| Alternative Parents | |
|---|
| Substituents | - Aminobenzoic acid or derivatives
- Benzoate ester
- 2-halobenzoic acid or derivatives
- Halobenzoic acid or derivatives
- Benzoyl
- Aniline or substituted anilines
- Chlorobenzene
- Halobenzene
- Aryl chloride
- Aryl halide
- Vinylogous halide
- Tertiary amine
- Tertiary aliphatic amine
- Amino acid or derivatives
- Carboxylic acid ester
- Carboxylic acid derivative
- Monocarboxylic acid or derivatives
- Organopnictogen compound
- Organic oxide
- Organohalogen compound
- Organochloride
- Organonitrogen compound
- Amine
- Organic oxygen compound
- Organooxygen compound
- Hydrocarbon derivative
- Organic nitrogen compound
- Primary amine
- Aromatic homomonocyclic compound
|
|---|
| Molecular Framework | Aromatic homomonocyclic compounds |
|---|
| External Descriptors | |
|---|
| Biological Properties |
|---|
| Status | Detected and Not Quantified |
|---|
| Origin | Exogenous |
|---|
| Cellular Locations | - Cytoplasm
- Extracellular
- Membrane
|
|---|
| Biofluid Locations | Not Available |
|---|
| Tissue Locations | Not Available |
|---|
| Pathways | | Name | SMPDB Link | KEGG Link |
|---|
| Chloroprocaine Pathway | Not Available | Not Available |
|
|---|
| Applications | |
|---|
| Biological Roles | Not Available |
|---|
| Chemical Roles | Not Available |
|---|
| Physical Properties |
|---|
| State | Solid |
|---|
| Appearance | White powder. |
|---|
| Experimental Properties | | Property | Value |
|---|
| Melting Point | 173-174°C | | Boiling Point | Not Available | | Solubility | 0.665 mg/mL | | LogP | 2.86 |
|
|---|
| Predicted Properties | |
|---|
| Spectra |
|---|
| Spectra | | Spectrum Type | Description | Splash Key | Deposition Date | View |
|---|
| Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (Non-derivatized) - 70eV, Positive | splash10-0k9i-9400000000-c227313b7fa92099b2dc | 2017-09-01 | View Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (Non-derivatized) - 70eV, Positive | Not Available | 2021-10-12 | View Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-014i-2590000000-d6a691c88bdf328bdece | 2016-08-03 | View Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-0gi0-2960000000-1227ef12643b1bc44f58 | 2016-08-03 | View Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-00di-9800000000-88711037c2b9e1e0a613 | 2016-08-03 | View Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-014i-0390000000-7684f7020aefa7770440 | 2021-10-11 | View Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-00fr-1910000000-dc231d66a4743a6fcd82 | 2021-10-11 | View Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-0059-5900000000-a2436cca9dd4087d6c5b | 2021-10-11 | View Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-0uk9-1590000000-3e4acb7ab260f903030c | 2016-08-01 | View Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-0udi-2930000000-1d40bf3d15aab91825f7 | 2016-08-01 | View Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-0umi-8900000000-4f493970e355eb62dd84 | 2016-08-01 | View Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-00di-0290000000-900e9b454cae510395ba | 2021-10-11 | View Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-0uk9-0930000000-ede990315b841dc52af0 | 2021-10-11 | View Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-0f89-2900000000-89a65d0b933b139cda9f | 2021-10-11 | View Spectrum | | MS | Mass Spectrum (Electron Ionization) | splash10-000i-9100000000-5a6841e384b0dfbd1238 | 2014-09-20 | View Spectrum |
|
|---|
| Toxicity Profile |
|---|
| Route of Exposure | Parenteral (intravenous injection). The rate of systemic absorption of local anesthetic drugs is dependent upon the total dose and concentration of drug administered, the route of administration, the vascularity of the administration site, and the presence or absence of epinephrine in the anesthetic injection. |
|---|
| Mechanism of Toxicity | Chloroprocaine acts mainly by inhibiting sodium influx through voltage gated sodium channels in the neuronal cell membrane of peripheral nerves. When the influx of sodium is interrupted, an action potential cannot arise and signal conduction is thus inhibited. The receptor site is thought to be located at the cytoplasmic (inner) portion of the sodium channel. It is hypothesized that Chloroprocaine binds or antagonizes the function of N-methyl-D-aspartate (NMDA) receptors as well as nicotinic acetylcholine receptors and the serotonin receptor-ion channel complex. |
|---|
| Metabolism | Chloroprocaine is rapidly metabolized in plasma by hydrolysis of the ester linkage by pseudocholinesterase.
Route of Elimination: Chloroprocaine is rapidly metabolized in plasma by hydrolysis of the ester linkage by pseudocholinesterase. Urinary excretion is affected by urinary perfusion and factors affecting urinary pH.
Half Life: 21 +/- 2 seconds |
|---|
| Toxicity Values | LD50: 97 mg/kg (Intravenous, Mouse) (1)
LD50: 950 mg/kg (Subcutaneous, Mouse) (1) |
|---|
| Lethal Dose | Not Available |
|---|
| Carcinogenicity (IARC Classification) | No indication of carcinogenicity to humans (not listed by IARC). |
|---|
| Uses/Sources | For the production of local anesthesia by infiltration and peripheral nerve block. They are not to be used for lumbar or caudal epidural anesthesia. |
|---|
| Minimum Risk Level | Not Available |
|---|
| Health Effects | Central Nervous System Effects: These are characterized by excitation and/or depression. Restlessness, anxiety, dizziness, tinnitus, blurred vision or tremors may occur, possibly proceeding to convulsions. High doses, or unintended intravascular injection, may lead to high plasma levels and related depression of the myocardium, hypotension, bradycardia, ventricular arrhythmias and, possibly, cardiac arrest. Neurologic side effects These observations may include spinal block of varying magnitude (including total spinal block), hypotension secondary to spinal block, loss of bladder and bowel control, and loss of perineal sensation and sexual function. Arachnoiditis, persistent motor, sensory and/or autonomic (sphincter control) deficit of some lower spinal segments with slow recovery (several months) or incomplete recovery have been reported in rare instances. [Wikipedia] |
|---|
| Symptoms | Not Available |
|---|
| Treatment | The first step in the management of convulsions, as well as underventilation or apnea due to unintentional subarachnoid injection of drug solution, consists of immediate attention to the maintenance of a patent airway and assisted or controlled ventilation with oxygen and a delivery system capable of permitting immediate positive airway pressure by mask. Immediately after the institution of these ventilatory measures, the adequacy of the circulation should be evaluated, keeping in mind that drugs used to treat convulsions sometimes depress the circulation when administered intravenously. Should convulsions persist despite adequate respiratory support, and if the status of the circulation permits, small increments of an ultra-short acting barbiturate (such as thiopental or thiamylal) or a benzodiazepine (such as diazepam) may be administered intravenously; the clinician should be familiar, prior to the use of anesthetics, with these anti-convulsant drugs. Supportive treatment of circulatory depression may require administration of intravenous fluids and, when appropriate, a vasopressor dictated by the clinical situation (such as ephedrine to enhance myocardial contractile force). If cardiac arrest should occur, standard cardiopulmonary resuscitative measures should be instituted. (3) |
|---|
| Normal Concentrations |
|---|
| Not Available |
|---|
| Abnormal Concentrations |
|---|
| Not Available |
|---|
| External Links |
|---|
| DrugBank ID | DB01161 |
|---|
| HMDB ID | HMDB15292 |
|---|
| PubChem Compound ID | 8612 |
|---|
| ChEMBL ID | CHEMBL1179047 |
|---|
| ChemSpider ID | 8293 |
|---|
| KEGG ID | C07877 |
|---|
| UniProt ID | Not Available |
|---|
| OMIM ID | |
|---|
| ChEBI ID | 3636 |
|---|
| BioCyc ID | Not Available |
|---|
| CTD ID | Not Available |
|---|
| Stitch ID | Chloroprocaine |
|---|
| PDB ID | Not Available |
|---|
| ACToR ID | Not Available |
|---|
| Wikipedia Link | Chloroprocaine |
|---|
| References |
|---|
| Synthesis Reference | Marks, H.C. and Rubin, M.I.; US. Patent 2,460,139; January 25,1949; assigned to Wallace
& Tiernan Products, Inc. |
|---|
| MSDS | Link |
|---|
| General References | - Wishart DS, Knox C, Guo AC, Cheng D, Shrivastava S, Tzur D, Gautam B, Hassanali M: DrugBank: a knowledgebase for drugs, drug actions and drug targets. Nucleic Acids Res. 2008 Jan;36(Database issue):D901-6. Epub 2007 Nov 29. [18048412 ]
- Drugs.com [Link]
- RxList: The Internet Drug Index (2009). [Link]
|
|---|
| Gene Regulation |
|---|
| Up-Regulated Genes | Not Available |
|---|
| Down-Regulated Genes | Not Available |
|---|